_______________________________________________________________
07.07.2020 - HIGH SENSITIVE COVID-19 REAL TIME PCR KIT- 80 MINUTES
The disease caused by the SARS-Cov2 virus and called COVID-19 has now caused a serious epidemic in the world and has been identified as a pandemic by the World Health Organization . Our company produced the CE approved COVID-19 Real Time PCR Kit, which detects the virus as 100% sensitive and 100% specific, which is the agent of COVID-19. The kit can give the results within 80 minutes. The sensitivity of the kit (Limit of Detection = LOD) is 1-10 copies / reaction.
Furthermore SNP Biotechnology company developed a Virus Inactivation & Transport Fluid for safe transportation of samples by inactivating enveloped and unenveloped viruses and a Viral RNA Extraction Kit which is highly sensitive (1-10 copy/ml), easy (within 25 min.), phenol-chloroform free, not need spin-colon method.
For detailed information of all these products, see the link below.
http://www.snp.com.tr/EN,38/covid-19.html
_________________________________________________________________
03.29.2021 SARS-CoV-2 VARIANT REAL TIME PCR KIT
In the autumn of 2020, several mutations were detected in SARS-CoV-2 different variants called UK Variant (B.1.1.7), South Africa Variant (B.1.351) and Brazilian variant (B.1.1.28) occured. Detection of variant types has become very important, as their contamination rate is high. With our two new products, you can easily detect SARS-CoV-2 variants.
Covid-19 Real Time PCR Kit v3 can detect RNA Dependent RNA Polymerase Gene (RdRP) and Nucleocapsid Protein Gene (N) regions of SARS-Cov-2 with high sensitivity and specificity. The kit also detect the N501Y mutation in the Spike Protein Gene (S) and determines whether the positive sample is variant or not.
With the SARS-CoV-2 Variant Real Time PCR Kit, a more detailed variant analysis can be performed. 69/70 Deletion and N501Y mutation in the Spike Protein Gene (S) can be detected from SARS-CoV-2 positive sample and can be determined whether it is a UK, South Africa or Brazilian variant or not.
For detailed information, please visit our COVID-19 page.

_________________________________________________
01.07.2020 - MEDLAB 2020
Our team is participating in Medlab 2020. We look forward to you visiting us in Dubai on 3-6 February. Our stand number is Z3.C19. Click on the link for more information.
https://www.omnia-health.com/exhibitor/snp-biyoteknoloji-arastirma-gelistirme-ve-uretim-san-ve-tic-ltd-sti

_______________________________________________________________
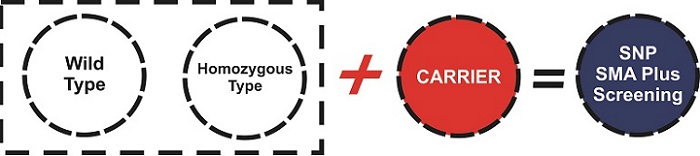
25.07.2019 - SMA PLUS SCREENING KIT
The Most Expeditious SMA Screening Kit
With the SMA Plus Screening Kit, you can screen SMA from both adults and newborns quickly, economically and reliably. You also can get the results in a practical way with the help of our special SMA Plus Software. Please click for detailed product list.
In addition, we developed SNP DBS DNA Extraction Kit specially for newborns therefore you can easily and easily isolate DNA from Guthrie paper.
_________________________________________________________
03.13.2019 - 2019 AHPL NEWBORN SCREENING
Our team is participating in 2019 APHL Newborn Screening and Genetic Testing Symposium in Chicago, IL. We look forward to you visiting us in Chicago on 7-10 April. Our stand number is 318. Click on the link for more information.
https://www.aphl.org/conferences/NBS-Symposiums/Pages/default.aspx

ADVANTAGES OF SMA PLUS SCREENING KIT
- Aim of the kit is giving the chance of early detection of carrier and homozygous SMN1 & NAIP within 70 minutes from blood and 85 minutes from DBS, which could meet the need of screening in the newborn babies and premarital couples rapidly and easily. By this way, it will be possible to provide early treatment and protection from neuronal loss seen within the first six months in infants with SMA.
- When SMN1 carrier infants are detected in the newborn screening program, their parents could be enrolled to pre-pregnancy screening. All identified carrier’s parents should be referred to follow-up genetic counseling for a discussion of risk to the fetus or future pregnancies.
- Identification of all carrier newborn babies and their carrier parents will reduce the burden of medical expenses for SMA diseases on the national budget. Prenatal and pre-implantation genetic diagnosis should be offered to the carrier couples. It gives the opportunity to decrease the number of SMA patients and treatment costs.
- It is also possible to detect the compound heterozygous for SMN1. The detected carriers with one deleted SMN1 allele will be sequenced to define whether or not the other allele is containing subtle intragenic mutations. By the way, the potential patients will be explored for SMA disease.
- Many studies have suggested that SMA patients with a deletion in NAIP show more-severe symptoms, but the direct role of NAIP has not been fully investigated.
- Detection of NAIP deletions, homozygous and/or carrier provides early warning about the deleterious effects on the neurological diseases.
____________________________________________________________
01.02.2019 - MEDLAB 2019
Our team is participating in Medlab 2019. We look forward to you visiting us in Dubai on 4-7 February. Our stand number is Z3.A10. Click on the link for more information.
https://www.omniagmd.com/exhibitor/snp-biyoteknoloji-arastirma-gelistirme-ve-uretim-san-ve-tic-ltd-sti

_________________________________________________________________
10.01.2018 - ASHG 2018
SNP Biotechnology is participating in American Society of Human Genetics 2018 Annual Meeting. We look forward to you visiting us in San Diego on 17-19 October. Our stand number is 1245. Click on the link for more information.
https://s23.a2zinc.net/clients/ASHG/ASHG2018/Public/eBooth.aspx?IndexInList=225&FromPage=Exhibitors.aspx&ParentBoothID=&ListByBooth=true&BoothID=100780
_________________________________________________________________
01.16.2018 - MEDLAB 2018
Our team is participating in Medlab. We look forward to you visiting us in Dubai on 5-8 February. Our stand number is Z3.F20. Click on the link for more information.
https://www.omniagmd.com/exhibitor/snp-biyoteknoloji-arastirma-gelistirme-ve-uretim-san-ve-tic-ltd-sti